Most commercial food products sold around the world require varying degrees of nutrition content and ingredient disclosure on their packaging. In countries where the regulations are more stringently codified, the…
Category: Genesis R&D Food
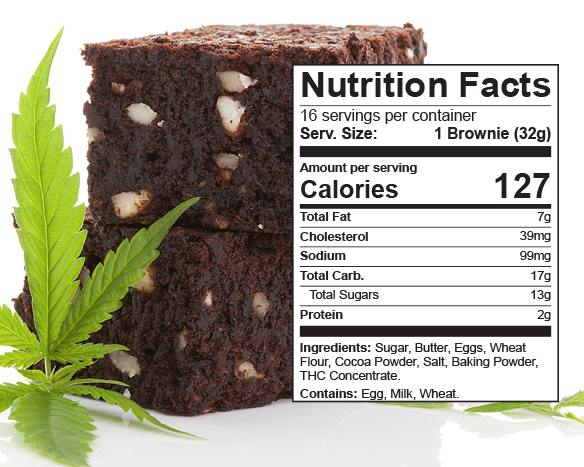
Cannabis Edibles Product Packaging and Compliant Labeling in Oregon
More and more states are legalizing the production and sale of edible cannabis products, both for medicinal and recreational use, and along with that comes regulations on how they must…

Tracking and Disclosing Sulfites in Genesis R&D Foods
The FDA requires a sulfite declaration on the packages of food products containing 10 ppm or more of sulfiting agents. Currently, there are six sulfiting agents allowed in processed foods:…

Best Practices for Database Naming Conventions
Defining naming conventions for your database can help keep your database organized and allow you to quickly filter and identify your Ingredients and Recipes. This post walks through best practices…

BE Disclosure vs. Non-GMO Labeling
There is some confusion about the difference between bioengineered (BE) ingredient disclosure and non-GMO labeling: What does each mean? What are the legal implications of both? What, if anything, does…

BE Disclosure Labeling FAQ's
Many of our customers have questions about the National Bioengineered Food Disclosure Law, which went into effect on Jan. 1, 2020 (January 1, 2021, for small food manufacturers). This blog…