Genesis R&D Foods Version 11.9 Adds Updates to Mexico Front-of-Package and Nutrition Facts Labels
Software program: Genesis R&D Foods
See also our tutorial on Creating a compliant Mexico Nutrition Facts Label.
The latest release of Genesis R&D Foods —Version 11.9 — was released in November 2020. Along with various minor software enhancements, this version incorporates the revisions and new features for complying with Phase 1 of the 2020 Mexico labeling regulation updates.
Watch our on-demand webinar where we cover all of these changes, Genesis R&D 11.9 Update: Changes to the Mexico Labeling Module. Additional details can be found in the software release notes.
This tutorial will cover the most notable changes:
New Mexico Nutrition Label Layouts
Label Templates in Genesis R&D Foods
The new Nutrition Facts Label formats have been incorporated into Genesis R&D. Updated label formats include:
- Standard/Vertical
- Linear
- Dual Column – Per Serving
- Dual Column – Per Container
To update your label to the 2020 regulations, open your Recipe, navigate to Edit Label and select “Mexico 2020” under category.
Overview of Layout Changes
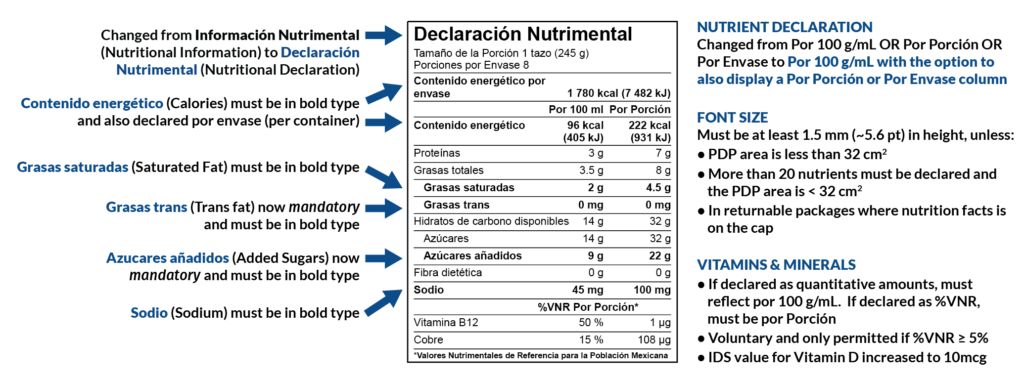
New Front-of-Package Warning Seals and Statements
Below is a brief overview of the Mexico Front-of-Package warning seals and statements. For more details on the specifics of these features and how they work in Genesis R&D Foods, check out this blog post.
Warning Seals
The Front-of-Package requirements and visuals have been completely redesigned. The original FOP icons were designed to inform consumers of the amounts of selected nutrients and proximates. In contrast, the new FOP icons alert consumers when the amounts exceed intake recommendations for Calories, Total Sugar, Saturated Fat, Trans Fat, and Sodium.
If one of those five nutrients exceeds the threshold amount, a warning icon will appear on your View Label page in Genesis R&D Foods. (Note: for the seals to appear for Saturated Fat and Trans Fat additional information will need to be provided on the Designations screen at the Ingredient level.)
If your recipe exceeds the threshold for all five nutrients, this is the image you would be required to place on your product packaging:

Cautionary Warning Statements
Caffeine
A warning statement “Contiene cafeína evitar en niños” (Contains caffeine, avoid giving to children) is required if the product contains any caffeine at all. This statement will automatically appear on the View Label screen if your recipe reports >0 caffeine in the “Caffeine” nutrient field.

Sweeteners
A warning statement “Contiene edulcorantes, no recomendable en niños” (Contains sweeteners, not recommended for children), is required on products containing excess sugar. For this statement to appear, additional information will need to be provided on the Designations screen at the ingredient level.
Nutrient Changes
Mandatory Nutrients
- Trans Fat is a new mandatory nutrient and is reported in mg.
- Added Sugars is a new mandatory nutrient and is reported in g.
Voluntary Vitamins and Minerals
- Vitamin B1, Vitamin B2, Vitamin B6, and Copper are now reported in µg instead of mg.
- The IDS value for Vitamin D has increased to 10mcg from 5,6.
- Voluntary vitamins and minerals will only appear on the label if the %VNR is 5% or greater (previously, a minimum of 5% was not required).
Other Nutrient Changes
Allulose, Polydextrose, and Tagatose have been added to the nutrients available in Genesis R&D. Allulose and Tagatose, when populated, will be used to calculate calories. Polydextrose can be displayed as a voluntary label nutrient.
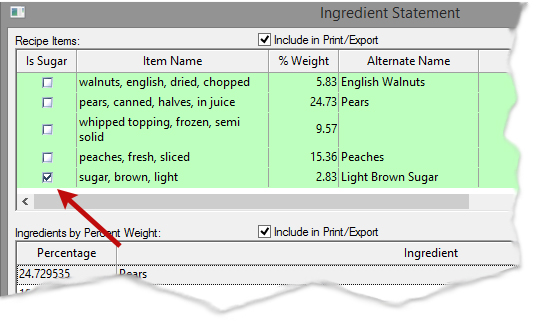
Ingredient Statement (Added Sugars Grouping)
Ingredients that are defined as sugars must now be grouped together on the ingredient statement. In order for this to work as designed, you have to designate the ingredient as sugar. This can be done on the Ingredient or Recipe levels.
Ingredients
When entering or editing an Ingredient, navigate to the Designations tab and select “Is Sugar.”

Recipes
When editing a Recipe’s Ingredient Statement, you will have the option to check “Is Sugar” next to each Ingredient in your Recipe.
Allergen Statement
- The allergen statement must be listed at the end of the list of ingredients in bold.
- Molluscs is a newly required allergen and must be declared in the allergen statement if the product contains molluscs.
- Tree nuts must be listed by specific type. Example: Almonds, walnuts, hazelnuts, pecans, etc.
- When contamination is possible during production, the label must use a “may contain” statement.
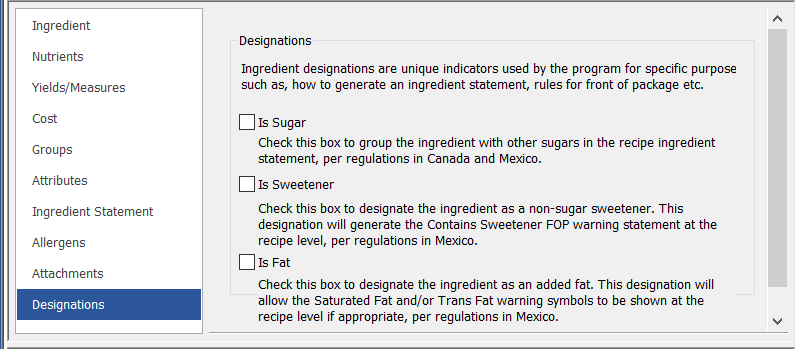
Designations
Ingredient Designations are used for compliance with the Mexico Front-of-Package and ingredient statement regulations as well as the ingredient statement regulations for Canada. There are currently three Designations options: Is Sugar, Is Sweetener, and Is Fat.
- Checking “Is Sugar” tells the program that this ingredient will be grouped with other sugars in the Ingredient Statement, per the labeling regulations for Mexico and Canada.
- Checking “Is Fat” or “Is Sweetener” lets Genesis R&D determine if an ingredient qualifies for a Mexico FOP warning icon or statement.
The Designations tab is a new section of the Ingredient record in Genesis R&D.
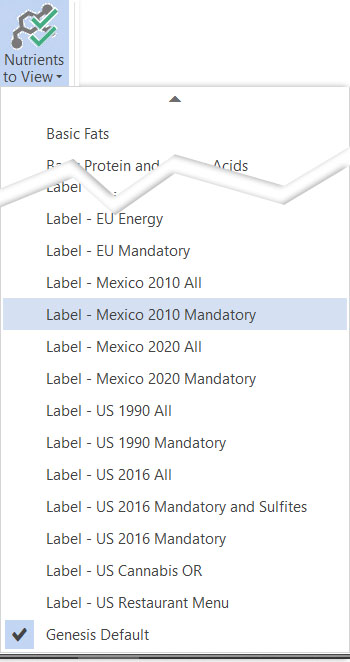
Nutrients to View
There are two new Nutrients to View sets:
- Label – Mexico 2020 Mandatory
- Label – Mexico 2020 All
Selecting one of these lets you easily view the Mexico-only nutrients in your reports or on your Recipe and Ingredient Nutrients page.
Nutrients to View options for Mexico Module.
Mexico Regulations Overview
To learn more about Mexico’s nutrition labeling regulation changes, check out the Mexico Labeling Updates blog, which covers these changes more extensively.
