The latest release of Genesis Foods, Version 11.13, is rolling out to customers this week. To continuously meet increasing regulatory demand, Genesis Foods will now include Phase 2 of the updated nutrition labeling regulations for Mexico and nutrition calculation changes for Canada.
In this blog, you will find updates related to:
- Global Software Updates
- Changes to the US Label Module
- Changes to the Mexico Label Module
- Changes to the Canada Label Module
- Additional Resources
Global Software Updates
- Improved high-resolution report display and printing: all formatting issues have been addressed and fixed across all releases for better reporting and visibility.
- Allow multiple recipes at a time to be added to Advanced Labels: Customers can now use ctrl + click to select multiple recipes from search results to add to your advanced labels.
- Improved handling of invalid protein scores for Label display: Previously, when an invalid protein score was added, labels displayed a 0 and in the new update will display a “–” instead.
Updates to the US Label Module
New Footnote Option Available for the Simplified Label
A new, shortened, footnote Format Option has been made available for the Simplified Label Display Type. This option allows the footnote to display a shortened version of the “Not a significant source of …” footnote that says, “other nutrients” instead of listing the individual nutrients by name.
To use this option Go to Edit Label > Format Options > U.S.
Note: this option can only be used when the label Display Type is set to Simplified

Updates to the Mexico Label Module:
Front-of-Package Phase 2
In accordance with the updated labeling regulations, made final on March 27, 2020, Phase 2 regulations have been implemented into Genesis Foods ahead of the compliance date to help customers get a head start on compliance. Implementation of Phase 2 begins October 1, 2023.
Mexico’s food labeling regulations require manufacturers to add warnings to their front-of-package (FOP) labels for foods and beverages high in calories, sugar, sodium, trans fat, or saturated fat, per the established parameters.
Key Changes to Note in Phase 2 Requirements
- Excess Calories Liquid: Warning symbol criteria: ≥ 70 calories total OR ≥ 8 calories from added sugars (was previously ≥ 10 calories from added sugars).
- Excess Sugars Liquid: Warning symbol criteria: beverages with <10 calories from added sugars are no longer exempt.
- Excess Sodium Solids: Warning symbol criteria: ≥ 1 mg sodium per calorie or ≥ 300 mg.
Additional Resources for Mexico FOP Regulations
Updates to the Canada Label Module
Updated Daily Value Calculations for Potassium and Sodium
On October 20, 2022, Health Canada amended the potassium and sodium Daily Values for specific age groups. These new Daily Values (DVs) impact the calculation of DVs for the Nutrition Facts table and specific nutrient content claims.
- Infants 6 to < 1 year: DV for potassium 860 mg (up from 700 mg)
- Children 1 to < 4 years: DV for sodium 1200 mg (down from 1500mg)
- Children 1 to < 4 years: DV for potassium 2000 mg (down from 3000 mg)
- All other age groups: DV for potassium 3400 mg (down from 4700 mg)
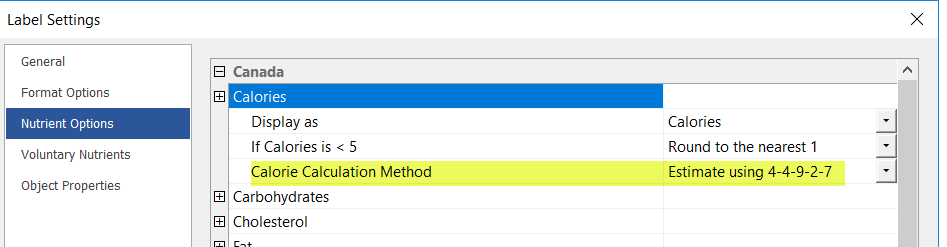
Calorie Calculation for Canada Updated to 4-4-9-2-7
Health Canada lists the average factors for energy values of protein, fat, carbohydrates, and alcohol with corresponding Cal/g. Genesis Foods has been updated to include the calories from alcohol when present.
The updated calculation should conclude as follows:
(4 x Protein) + (9 x Fat) + (4 x [Carbs-Dietary Fiber]) + (2 x Dietary Fiber) + (7 x Alcohol)
Note: The default Calorie Calculation Method for Canada has been updated to use Estimate using 4-4-9-2-7. As a result, all newly created Recipes will use this calorie calculation method by default.
To view or update the default calculation on existing Recipes, go to Edit Label > Nutrient Options > Calories > Calorie Calculation Method
Read the Full Release Notes
For more information on the technical updates for this release, check out our Release Notes, or reach out to support@esha.com for questions.
Other posts you might be interested in
View All Posts
Food Labeling
3 min read
| April 9, 2019
Considerations for Implementing the FDA’s Restaurant Menu Labeling Laws
Read More
Food Labeling
4 min read
| December 14, 2016
Health Canada Finalized Food Labelling Regulations
Read More
Trustwell News
3 min read
| October 4, 2022

